Potential to Simplify the Writing of Submission Documents: Evaluation of Publicly Available Module 2 Documents in Drug Submissio
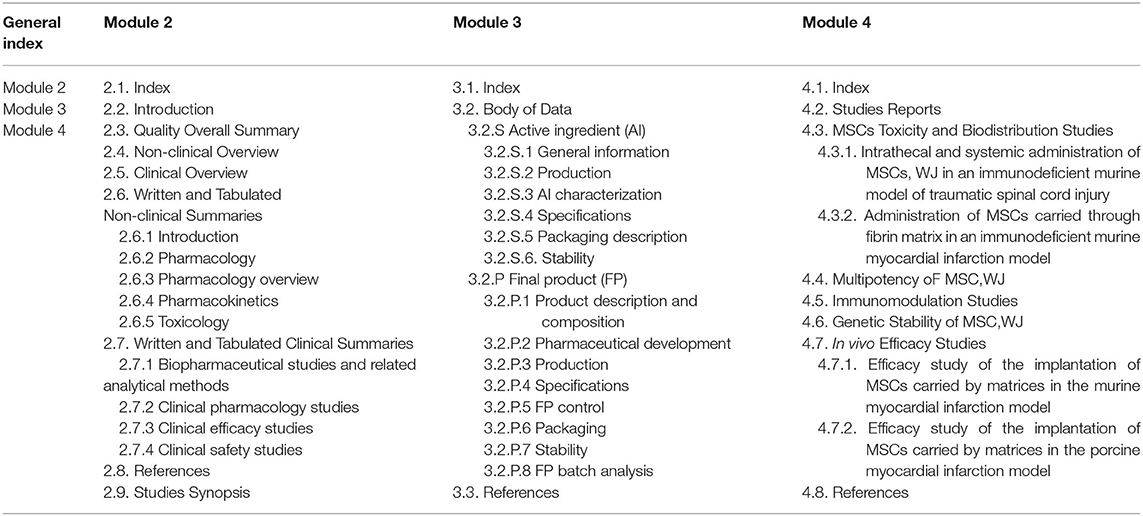
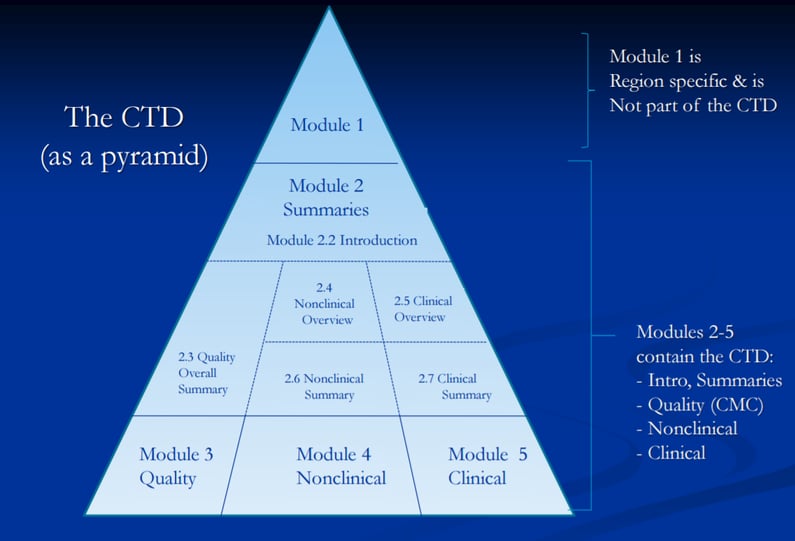
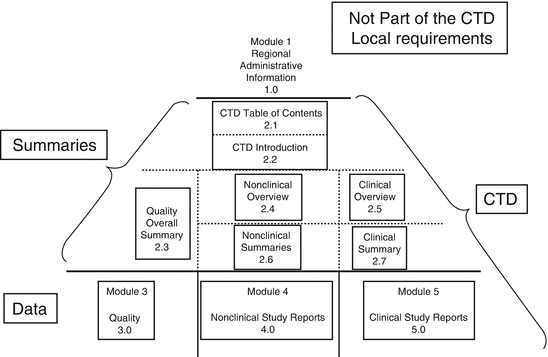
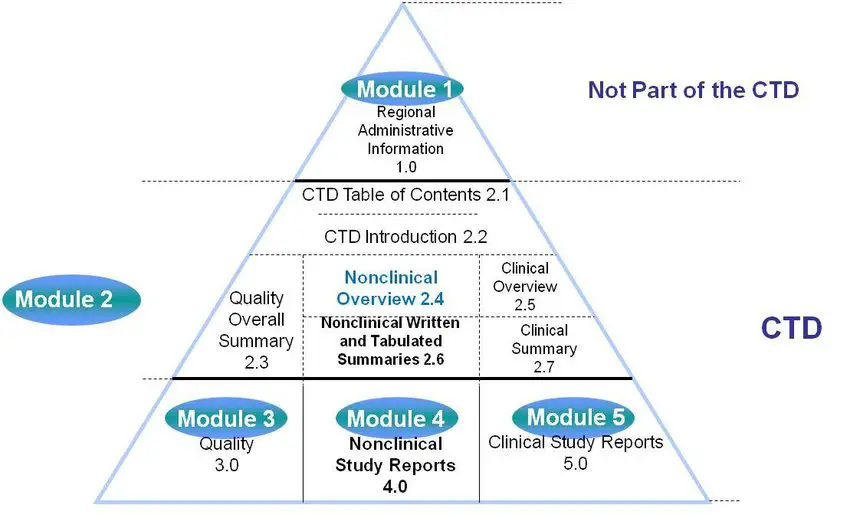
Clinical Study Reports Quality 2.7 Clinical Summaries 2.5 Clinical Overview 2.3 Quality Overall Summary 22.6 Non-Clinical Summar
WHO Guidelines on submission of documentation for the pilot procedure for prequalification of similar biotherapeutic products fo

PDF) The Preparation of a Preclinical Dossier to Support an Investigational New Drug (IND) Application and First-in-Human Clinical Trial
![PDF] Abbreviated clinical study reports with investigational medicinal products for human use: current guidelines and recommendations. | Semantic Scholar PDF] Abbreviated clinical study reports with investigational medicinal products for human use: current guidelines and recommendations. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7b16b80697ade631fb500770299d798221438a17/2-Figure1-1.png)
PDF] Abbreviated clinical study reports with investigational medicinal products for human use: current guidelines and recommendations. | Semantic Scholar