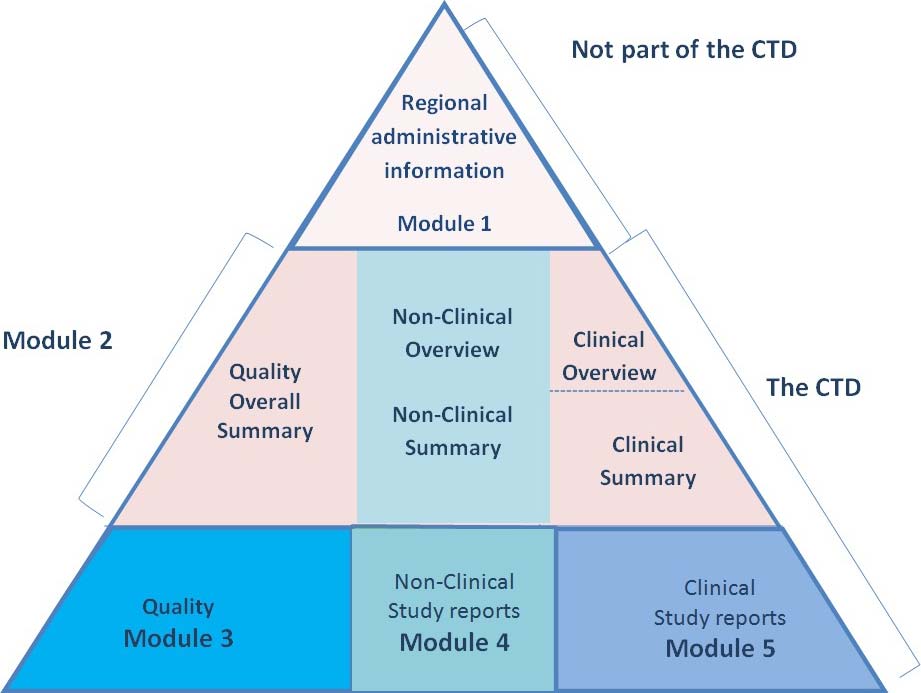
The Future of CMC Regulatory Submissions: Streamlining Activities Using Structured Content and Data Management - ScienceDirect
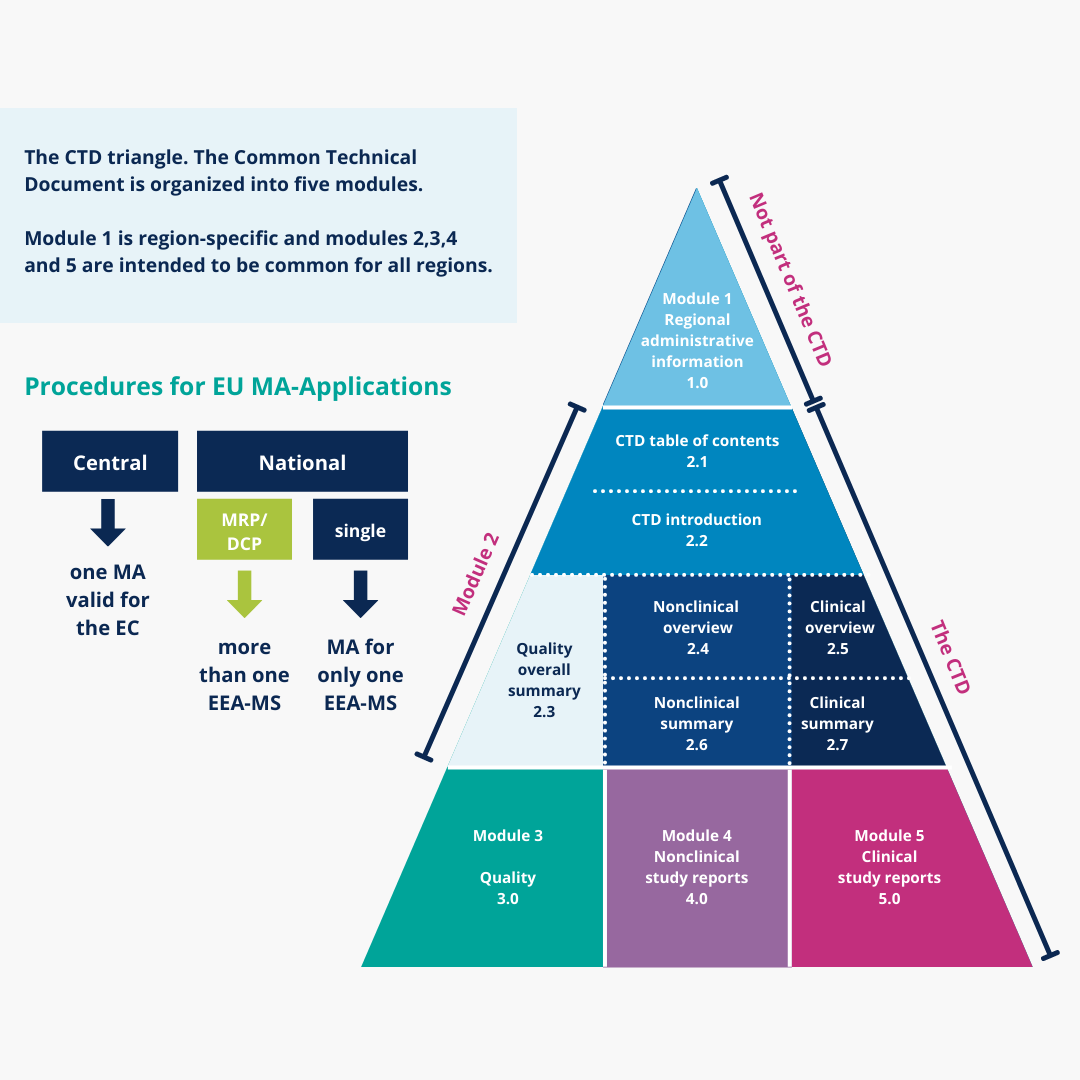
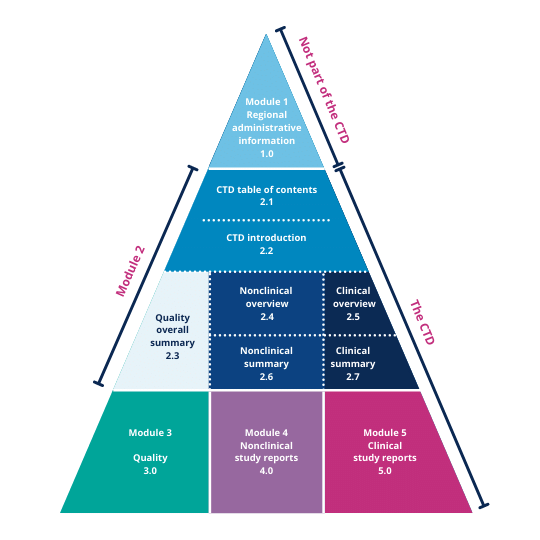
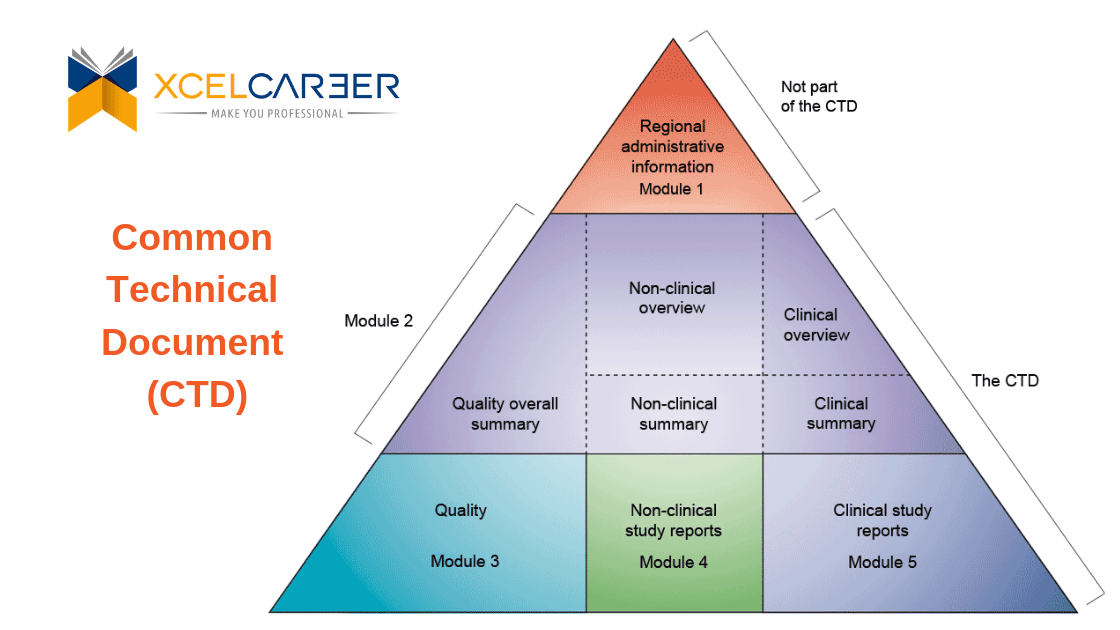
Understanding the Five Modules of the CTD Format in Regulatory Affairs for Pharmaceutical Submissions"

PDF) Preparation and Review of Chemistry, Manufacturing and Control (CMC) Sections of CTD Dossier for Marketing Authorization | Dhruvi Patel - Academia.edu
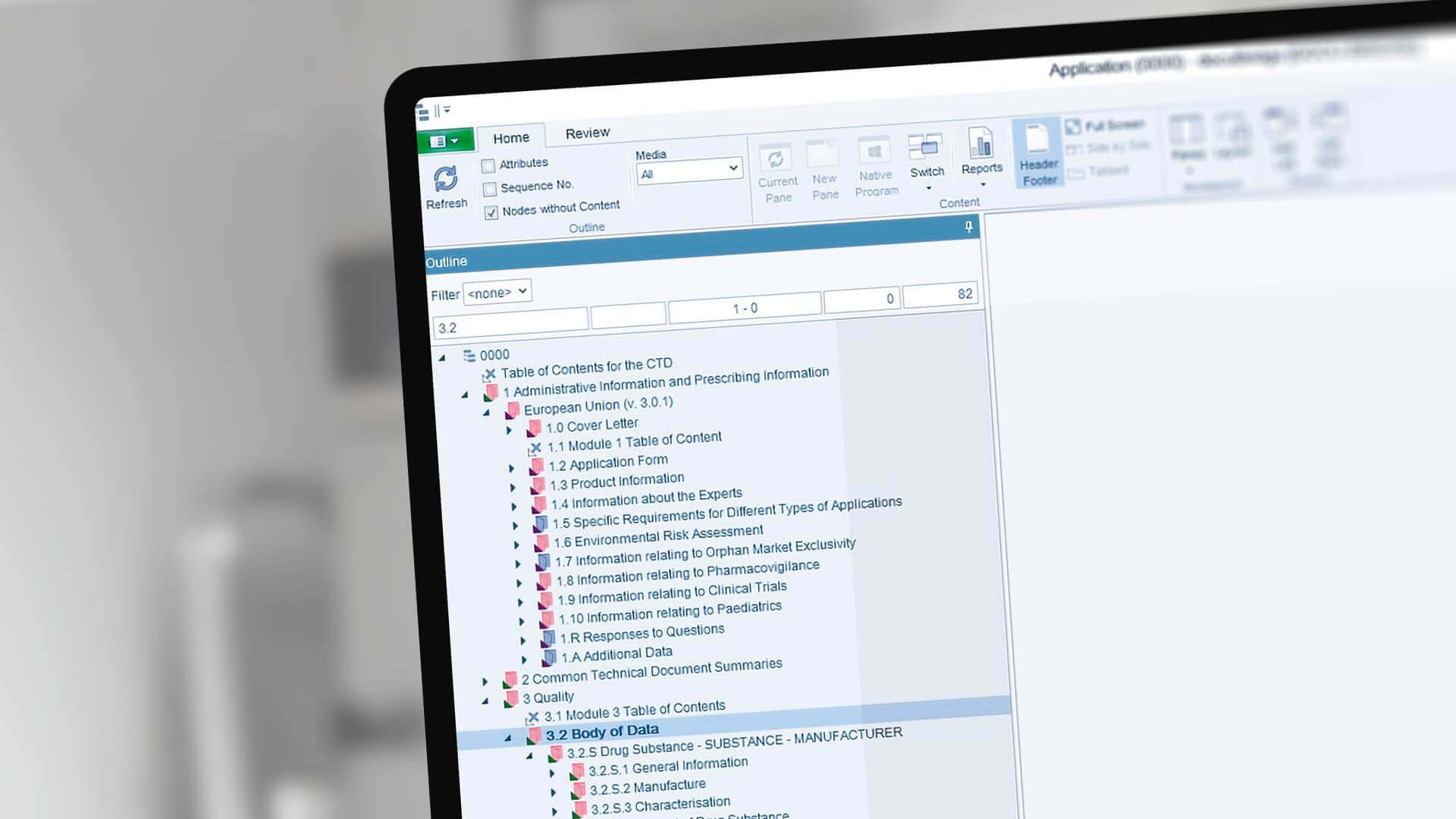
Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substan

The Organisation for Professionals in Regulatory Affairs - TOPRA - LAST CHANCE! Now available ONLINE How to effectively prepare and manage module 3 of the dossier covering pharmaceutical (CMC) information. Get up

MASTERCLASS LEGAL & REGULATORY AFFAIRS Aus der Praxis - für die Praxis Modul 3 - Gestaltung der Verpackung

Regulatory Affairs Explained Series Episode 5 | Module 3 - Chemistry, Manufacturing & Controls (CMC) - YouTube




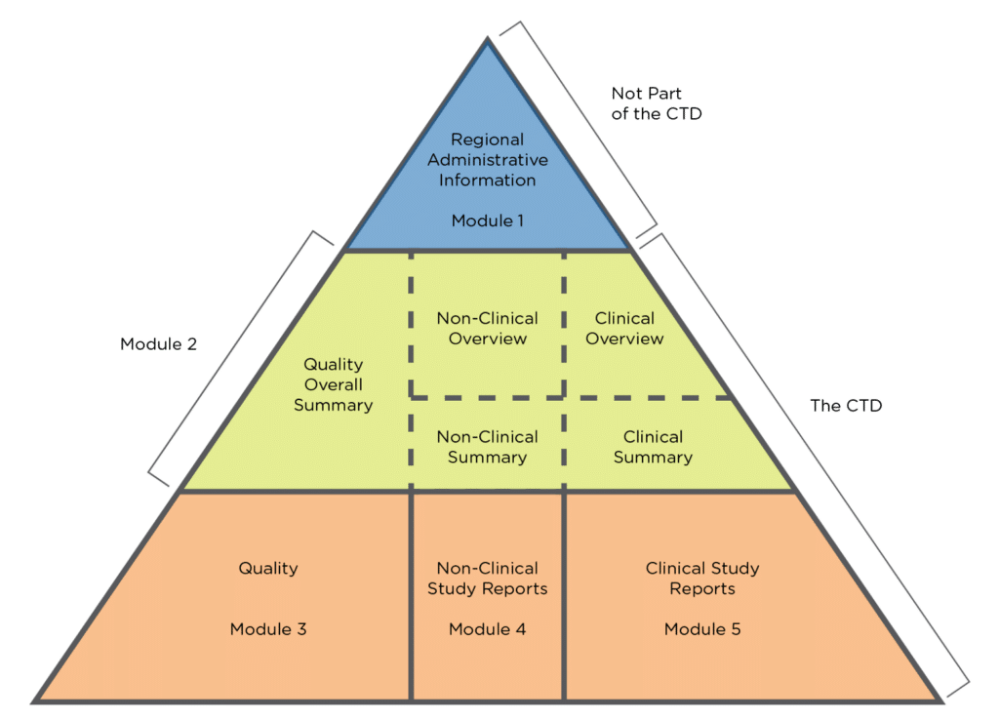
(1).png)