Combination denosumab and high dose teriparatide for postmenopausal osteoporosis (DATA-HD): a randomised, controlled phase 4 trial - The Lancet Diabetes & Endocrinology

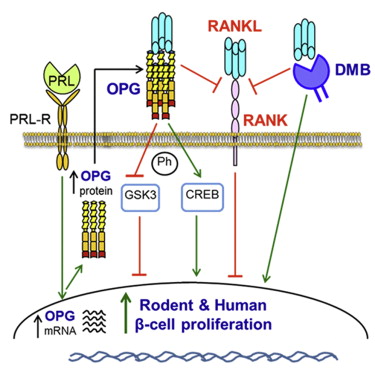
Denosumab in postmenopausal women with osteoporosis and diabetes: Subgroup analysis of FREEDOM and FREEDOM extension - ScienceDirect

iSpatula - The US Food and Drug Administration (FDA) has approved denosumab (Prolia, Amgen) for the treatment of glucocorticoid-induced osteoporosis in men and women at high risk of fracture, defined as defined

Denosumab and incidence of type 2 diabetes among adults with osteoporosis: population based cohort study | The BMJ

Combination denosumab and high dose teriparatide for postmenopausal osteoporosis (DATA-HD): a randomised, controlled phase 4 trial - The Lancet Diabetes & Endocrinology