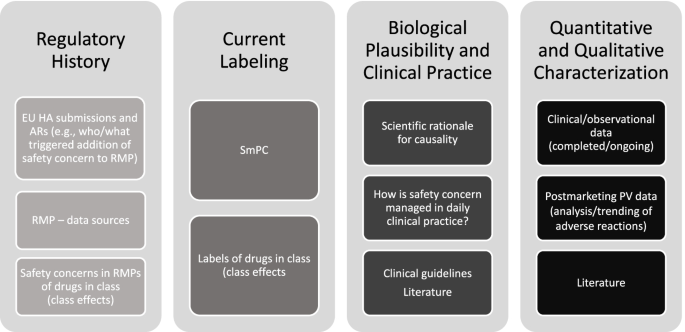
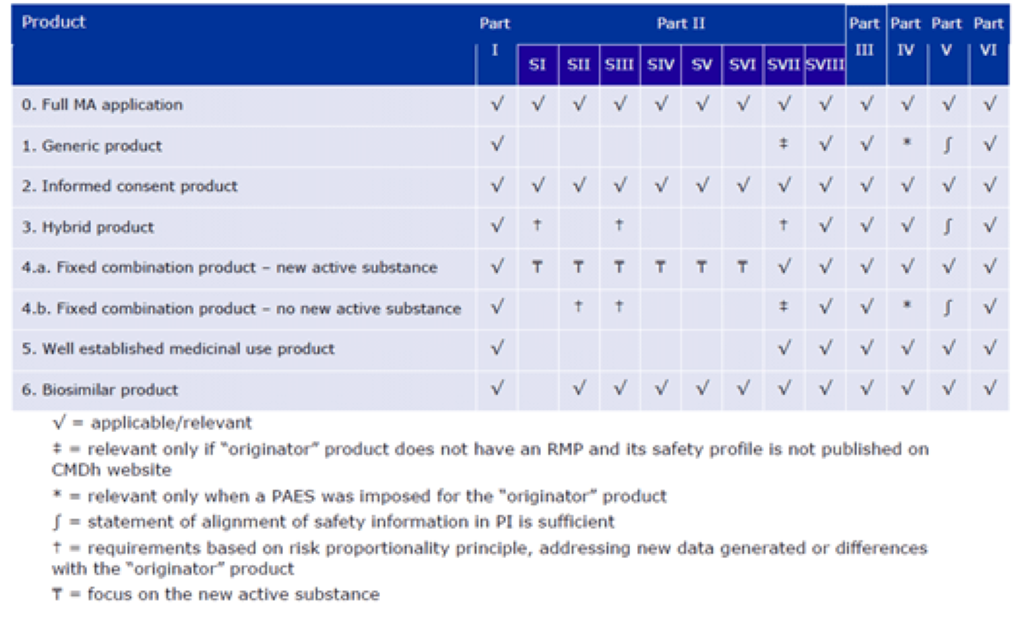
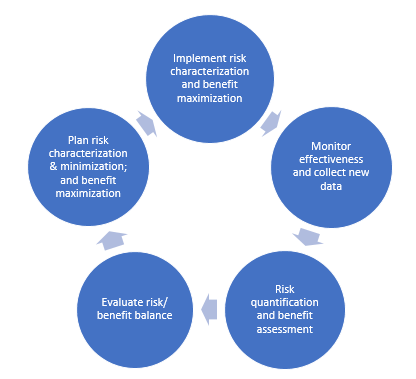
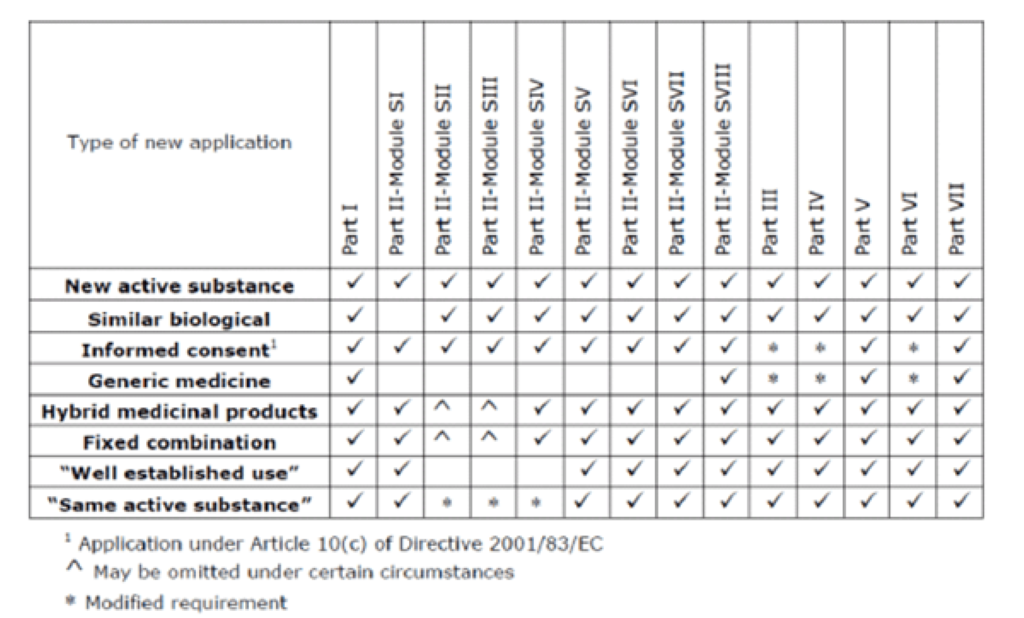
PDF) Response to European Medicines Agency Consultations on 'Good Pharmacovigilance Practice' (GVP Module V) and 'Guidance on format of the risk management plan' (RMP)
Risk Management Plans: reassessment of safety concerns based on Good Pharmacovigilance Practices Module V (Revision 2)—a company experience | Journal of Pharmaceutical Health Care and Sciences | Full Text

Interview Question: Pharmacovigilance Aggregate reporting How many Parts and modules are there in RMP? Answer: RMP consists of 5 parts and 8 modules under part 2. #interview #questions #aggregatereports #PV #pharmacovigilance... -